Title: Common things are common, rare things are rare – A 66-year-old patient with a newly diagnosed AML presenting with a saprophytic fungus
Submitted by: Jana Wittig
Institution: University Hospital Cologne
Email: jana.wittig@uk-koeln.de
Date Submitted: 2021–11–26
History:
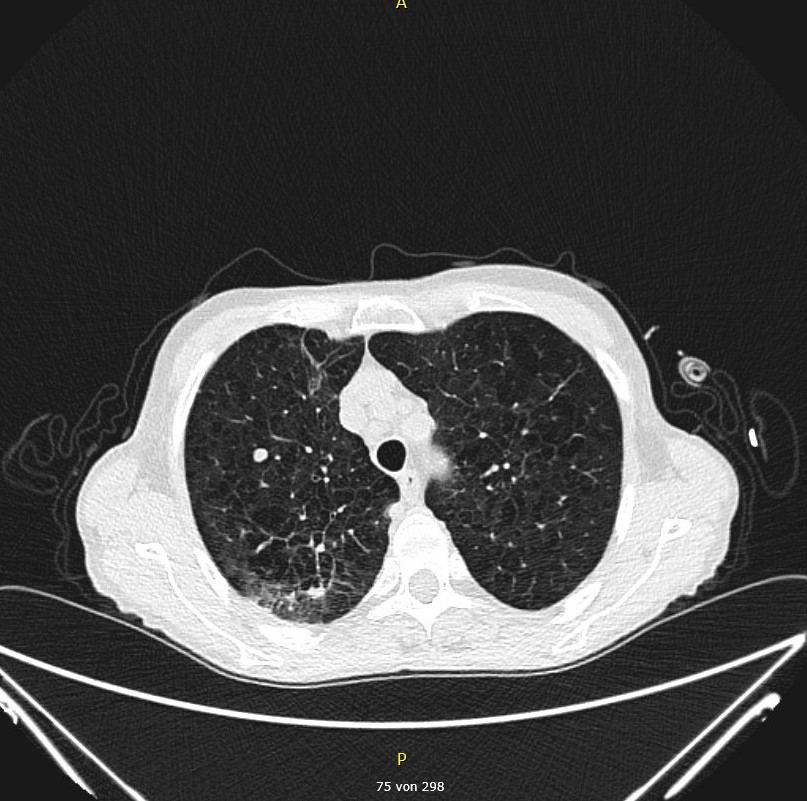
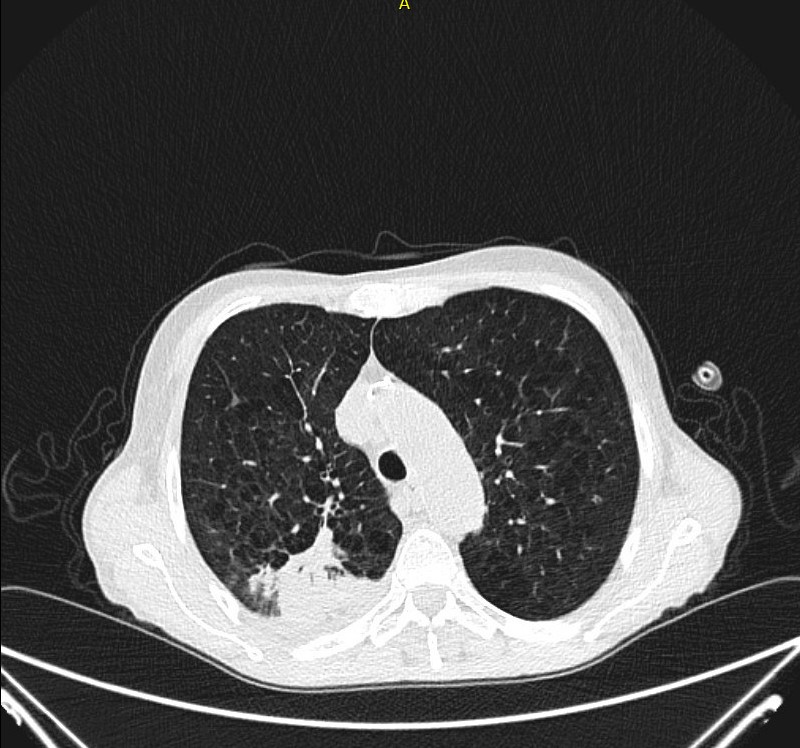
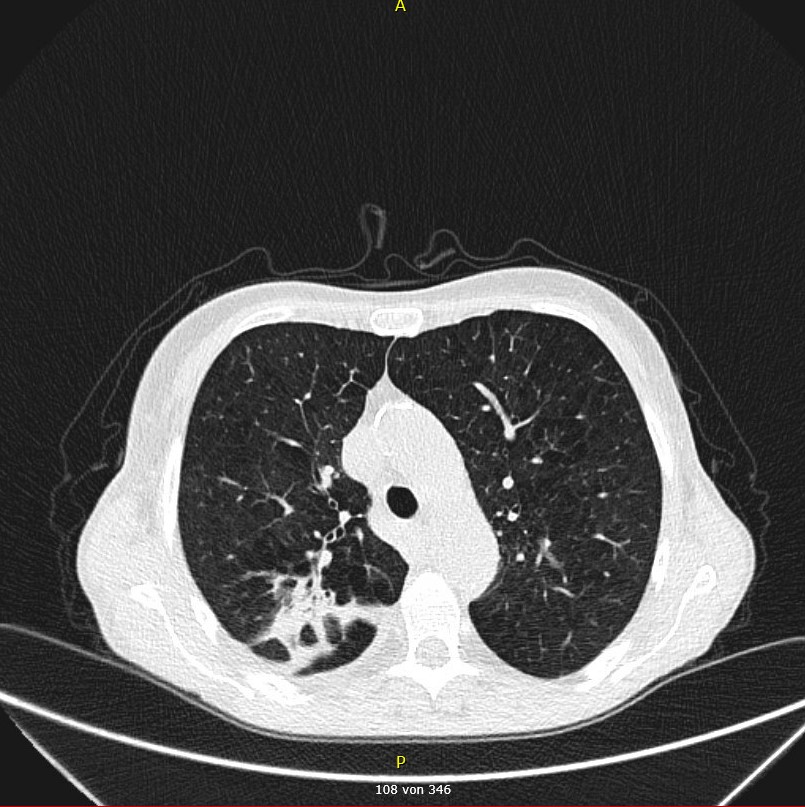
A 66-year-old woman presented with exertional dyspnea, leg edema, pretibial skin hemorrhages and subjectively perceived deterioration of her general condition. The medical history included hypothyroidism. The peripheral blood analysis at admission to hospital showed pancytopenia (Hgb 8.4 g/dl, WBC 3.03 x 10*9/l, platelets 28 x 10*9/l) as well as the presence of 46% of blasts. The results of the bone marrow puncture on the following day confirmed the diagnosis of acute myeloid leukemia (AML). The baseline CT of the thorax five days after admission to hospital showed a solid round focus (Fig. 2) and a dorsobasal infiltrate within the right lung lobe (Fig. 3).
Physical Examination on admission to hospital:
On physical examination, the patient had multiple petechiae and extensive skin hemorrhages spread over the entire integument and lower limb edema of both legs. Auscultation of the heart and lungs was unremarkable.
Vital Signs on admission to hospital:
BP 130/80, HR 90, O2 Sat 98% RA, RR 20, T 37.4°C
Laboratory Examination on admission to hospital:
Pancytopenia: RBC 2.60 x 10*12/l, Hgb 8.4 g/dl, HCT 24%, WBC 3.03 x 10*9/l, platelets 28 x 10*9/l, neutrophils: 0.16 x 10*9/L, lymphocytes: 0.91 x 10*9/l
Question 1: What are probable/possible diagnoses?
The most likely differential diagnoses for the pulmonary infiltrate in the right lobe of the lung were 1) an infectious cause such as invasive mycosis or tuberculosis, 2) a secondary malignancy, 3) scar tissue or 4) a benign tumor such as hamartoma. Since the patient suffered from functional aplasia regarding the newly diagnosed AML, opportunistic infections had to be considered.
The round focus detected in the right lung lobe on the baseline CT remained constant and unchanged in all subsequent CT examinations, so that it will not be described further below due to its lack of relevance for this case (Fig. 2).
Diagnostic Tests/Microbiology Performed:
- PET-CT scan: No indication of second pulmonary malignant formation (no pathologic FDG-uptake), round focus suspicious of infectious cause, most likely invasive fungal disease.
- Blood cultures: No pathological finding
- Aspergillus serological testing: No pathological finding
- PCR testing in BAL for Mycobacterium tuberculosis, Mycoplasma pneumoniae, Chlamydia pneumoniae, Legionella pneumoniae, Aspergillus species, Mucorales, Pneumocystis carinii, Toxoplasma gondii: Negative
- Bronchoalveolar lavage: Cultural identification of Peniophoraceae species
Final Diagnosis:
Systemic pulmonary mould mycosis caused by Peniophoraceae species
Question 2: What treatment is recommended in the care of this patient?
Treatment:
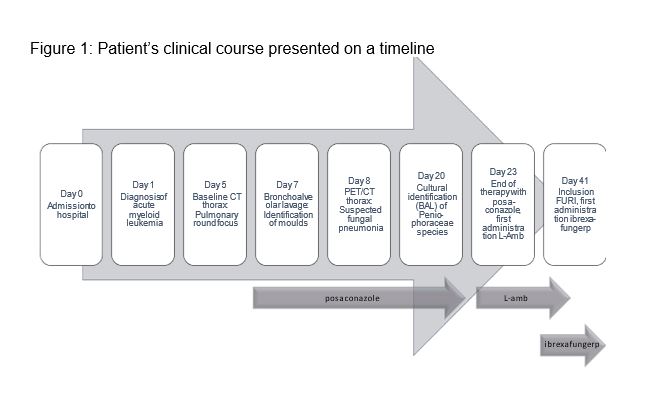
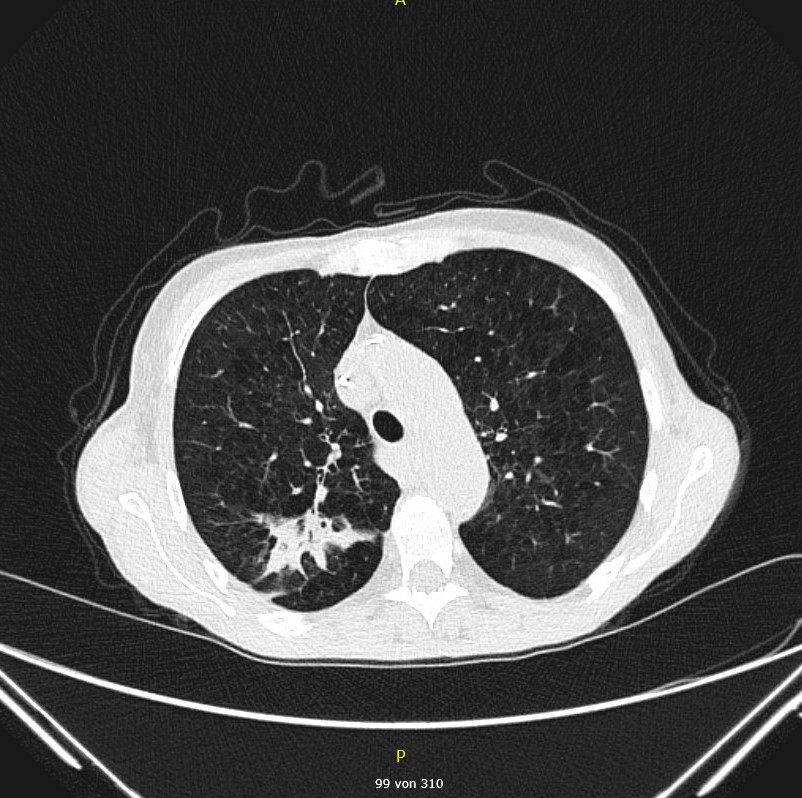
We initiated antifungal prophylaxis with posaconazole (300 mg/d, tablet for oral intake) five days after admission to hospital (Fig. 1). After the bronchoalveolar lavage tested positive by culture and revealed the colonization with moulds, we continued treatment with posaconazole at the same dosage. For the treatment of the newly diagnosed acute myeloid leukemia (AML) an induction therapy according to the 7+3 regimen (cytarabine and daunorubicin) supplemented by GO (gemtuzumab and ozogamicin) was administered for the first time 12 days after admission to hospital. During the administration of posaconazole, we observed disease progression in radiological follow-up examinations and therefore replaced posaconazole with liposomal amphotericin B (3 mg/kg BW/d, infusion solution) after 15 days of treatment (Fig. 1). For technical reasons an inclusion into the OPAT-programme was not possible. OPAT is defined as the administration of a parenteral antibiotic treatment in the outpatient setting. Since this patient did not agree on ambulatory parenteral administration of liposomal amphotericin B, the therapy could not be continued. Other effective antifungal therapeutic options for oral intake did not exist at that point. We therefore decided to initiate a therapeutic attempt with ibrexafungerp (250 mg tablets at a dose of 750 mg bid) within the framework of the FURI study (Fig. 1). Up to the present day, the regular CT follow-ups have shown stable to regressive pulmonary infiltrates, i.e., a good response to antifungal treatment (Fig. 4 and 5).
Outcome:
The regular clinical and radiological follow-ups have shown a satisfactory treatment response after initiating the antifungal therapy with ibrexafungerp (Fig. 4 and 5). The bone marrow puncture 16 days after first administration of chemotherapy showed a complete hematologic remission. Consolidation therapy was continued with high dose cytarabine (HiDAC) and is up until now still ongoing.
Discussion:
Peniophoraceae species is a family of saprophytic fungi, that are typically found in the plant kingdom on the bark of trees. They are also known as wood-decay fungus, causing white rot. Up until now, Peniophoraceae have not been repeatedly identified as a pathogen in humans. Only an individual case of a farmer infected with Peniophoraceae has been reported (Miranda et al.). In that case report, the patient was previously exposed to specific work-related environmental conditions, which was not the case in our patient. Patients in functional aplasia due to an underlying hemato-oncological disease are known to be particularly susceptible to invasive fungal infections like pulmonary aspergillosis and invasive candidiasis (Ruhnke et al.). Since there is no established therapy for Peniophoraceae, we initially continued the antimycotic therapy with posaconazole because of its concurrent therapeutic efficacy against moulds. After observing a clinical progress of the disease, we escalated the antimycotic therapy by replacing posaconazole with liposomal Amphotericin B. Initially we were hesitant to initiate therapy with ibrexafungerp, since peniophoraceae do not produce conidia and resistance testing was therefore not feasible. Since the patient refused to participate in the APAT programme, we had to stop therapy with parenteral liposomal amphotericin B. In the absence of further effective and orally available antimycotic drugs for this patient, we eventually opted for therapy with ibrexafungerp within the FURI study (SCY-078). Ibrexafungerp is an oral glucan synthase inhibitor, that is approved for the treatment of vulvovaginal candidiasis only (Masone et al.). FURI (SCY-078) is an open-label study investigating effectiveness and safety of ibrexafungerp in patients with mycotic disease, who are either refractory to treatment or intolerant towards the administered antimycotic therapeutic agents. Our patient received the first dose of ibrexafungerp 20 days after the identification of Peniophoraceae species. The CT-based follow up scans at one week and at eight weeks after the first ibrexafungerp dose showed regression and in the further course of time, a stability of the previously described mycotic infiltrates (Fig. 4 and 5). This can be considered a good therapeutic response to antifungal therapy. In regular clinical evaluations, we have also recorded an improvement of the patients’ overall clinical status.
Chronological development of the morphological findings in baseline and follow-up CT
Day 5: Baseline CT before initiation of antifungal therapy shows
- Perihilar solid round focus in the right upper lobe (CT Scan 1) and
- Infiltrate in the dorsobasal right upper lobe (CT Scan 2)
Key References:
L. Miranda, O. Epelbaum, H. Assallum, and G. Miranda. Moldy Hay and the Cirrhotic Farmer: The First Case of Peniophora Lung Infection C59. UNUSUAL PULMONARY INFECTIONS. May 1, 2018, A5467-A5467
Gou Q, Ren C, Peng C. Characterization of the complete mitochondrial genome of Peniophora lycii (Russulales: Peniophoraceae) with its phylogenetic analysis. Mitochondrial DNA B Resour. 2021 Jul 5;6(8):2200-2202. doi: 10.1080/23802359.2021.1945508. PMID: 34286082; PMCID: PMC8266238.
https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&id=103393&lvl=3&lin=f&keep=1&srchmode=1&unlock [Last access 11-18-2021, 11:30 p.m.]
Ruhnke et al. Treatment of invasive fungal diseases in cancer patients—Revised 2019 Recommendations of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Oncology (DGHO). Mycoses. 2020 Feb. DOI: 10.1111/myc.13082
Masone, M.C. Ibrexafungerp to treat acute vulvovaginal candidiasis. Nat Rev Urol 18, 638 (2021). https://doi.org/10.1038/s41585-021-00522-9
https://clinicaltrials.gov/ct2/show/NCT03059992 [Last access 11-18-2021, 12 p.m.]
Barr DA, Seaton RA: Outpatient parenteral antimicrobial therapy (OPAT) and the general physician. Clin Med 2013;13(5):495-499. doi:10.7861/clinmedicine.13-5-495
