Title: Invasive aspergillosis due to A. fumisynnematus: an emerging pathogen in an immunosuppressed patient
Submitted by: Daniel Aguilar-Zapata
Institution: Hospital Médica Sur. Mexico City, Mexico
Email: daniel_aguilar@hotmail.com
Date Submitted: 01/02/2025
History:
A 77 year old immunosuppressed man from the south area of Mexico City, an area with significant humidity, presents with fever and cough. Patient reports that “the walls of the closets and attic of the house have damp areas”. The patient admits that he performed work in the attic of his house intermittently, even with his underlying condition. He has a diagnosis of myelodysplasic syndrome, and before his symptoms emerged. He reports being treated with azacitidine (Vidaza®) during 10 months followed by cyclophosphamide plus eltrombopag one month before the rising symptoms.
PHYSICAL EXAM, ASSESSMENTS:
Main complaint: fever of 5 days with a productive cough with yellowish sputum. Also his wife noted the presence of a firm, motile, painful nodules localized in the left arm (1.3 x 1.3 inches) and his right thigh (1.2 x 2 inches).
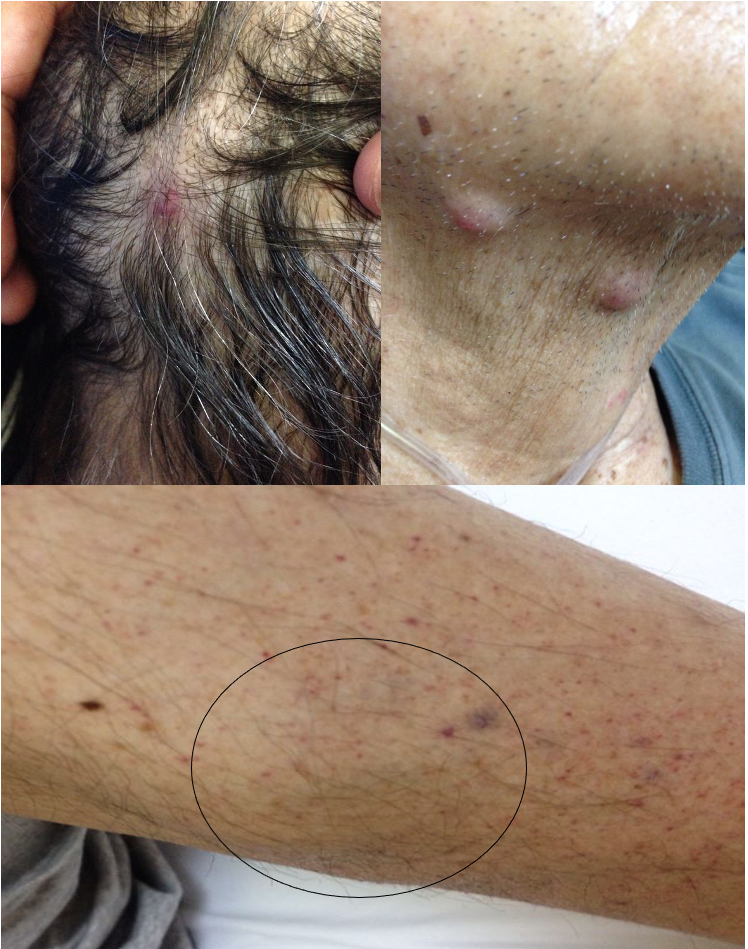
At first they related it to the petechiae that appeared indistinctly on the body. In the following days new nodules appeared in the scalp, neck, legs, torso and back (those had a smaller size, around (0.20 x 0.40 inches each). We notice that some of the nodules were erythematous with ulcer in the middle (figure 1).
An infectious disease consult was called to differentiate whether the respiratory symptoms were related to the skin lesions that were appearing in the setting of a febrile syndrome in an immunosuppressed patient.
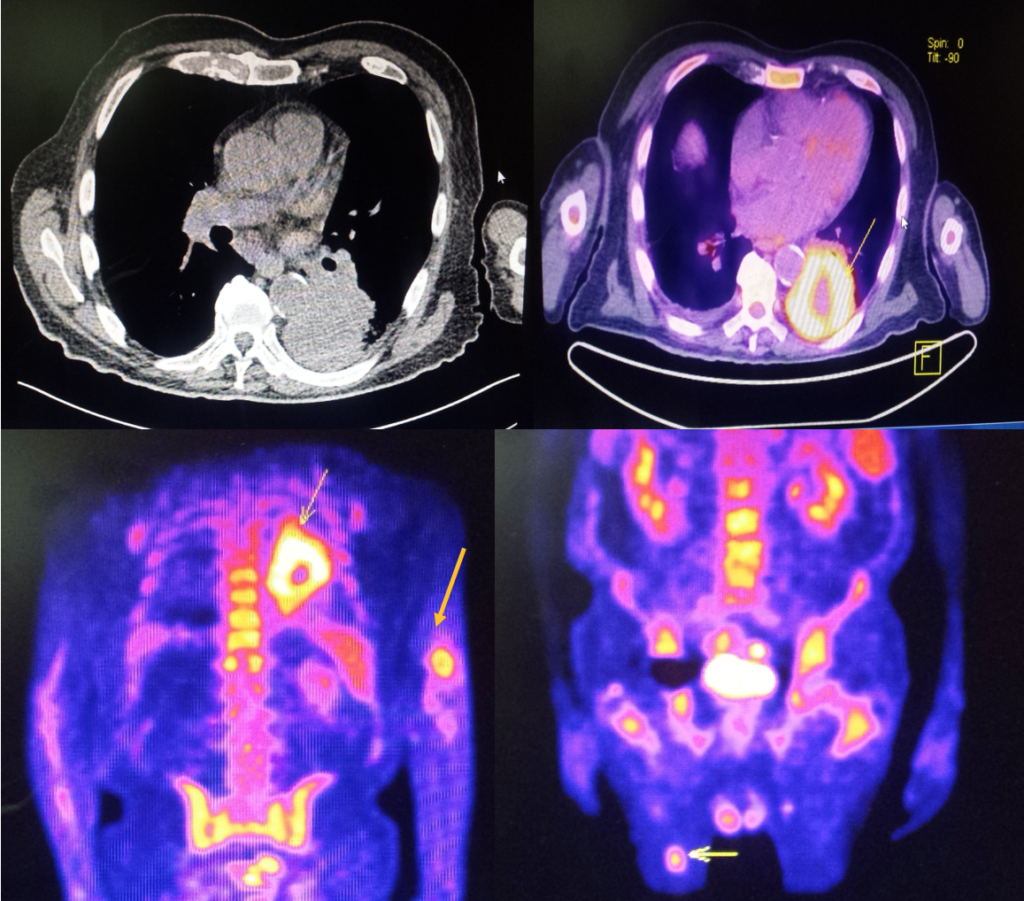
A full body CT-PET scan was performed demonstrating metabolic activity in lung consolidation, and in the nodules from the left arm and right thigh. A biopsy was performed in those nodules on the left arm and right thigh (figure 2).
We also recommended that a bronchoscopy with bronchoalveolar lavage, and a lung biopsy be performed, as oncology wanted to also rule out solid tumor in the lung verses infection. .
Laboratory Examination:
Hemoglobin: 10 g/dL; Platelets: 2,000 x103/μL; WBC’s 3.0 x103/μL, Neutrophils: 1.3 x103/μL, Lymphocytes 1.1 x103/μL, Eosinophils 0.4 x103/μL
Question 1: What are probable/possible diagnoses?
Microbiology/Diagnostic Tests Performed:
BAL Galactomannan: 0.63 ; Serum galactomannan 0.61
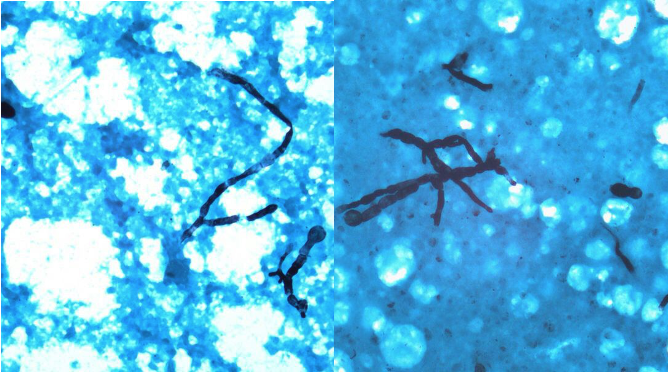
Nodule biopsy (right thigh): septate hyphae with acute angle branching (figure 3).
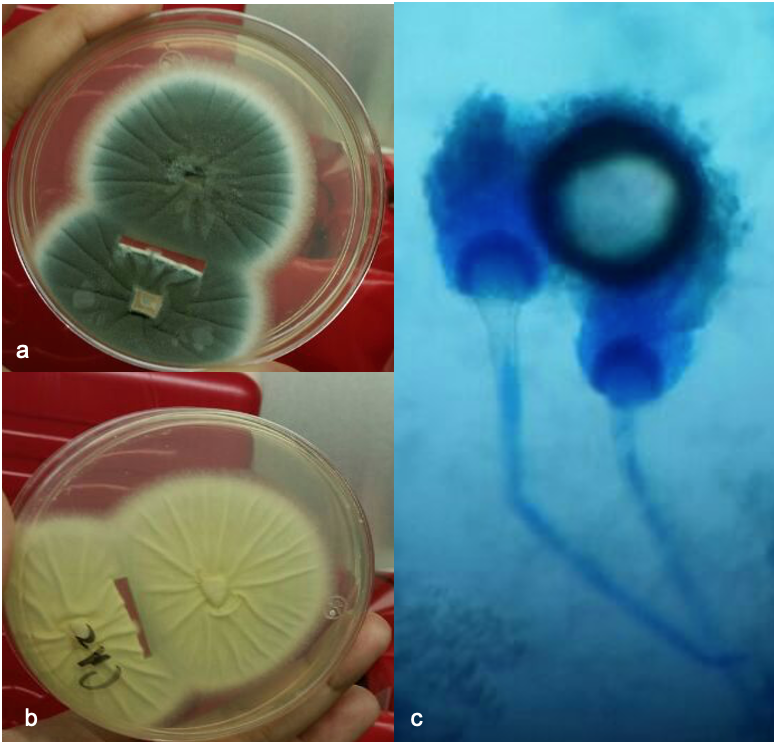
BAL cultures and nodule biopsy: on sabouraud broth a small, graygreen color with a cottony texture colonies, cotton blue smear revealed septade hyphae with a finely roughened conidia morphologically identified as A. fumigatus (figure 4a, 4b).
Lacking MALDI-TOF MS at the time, we took the sample to molecular identification, using DNA extraction and PCR to identify the Aspergillus species as follows Genomic DNA was isolated using a DNeasy blood and tissue kit (Qiagen, Ventura CA, USA) according to the manufacturer’s instructions. Molecular identification of isolates from three cases was achieved by 18S-ITS1-5.8S-ITS2-28S rRNA gene amplification and sequencing, using a set of primers ITS1-5’-TCCGTAGGTGAACCTGCGG-3’ and ITS4-5’-TCCTCCCGCTTATTGATATGC-3’ *. The PCR reactions contained 25 ml of Top Taq master mix (Qiagen, Ventura CA, USA), 10 ng of DNA (10 ml), and 25 mM of each primer (2 ml) in a total volume of 50 ml. PCR conditions were: 30 cycles of denaturing (94 °C, 1 min), annealing (58.5 °C, 1 min) and extension (72 °C, 1 min), followed by a final extension at 72 °C for 10 min. A sample of 10 ml of product from each PCR was electrophoresed in a 1.5% agarose gel with 0.5 mg of ethidium bromide/ml and 1X Tris-acetate-EDTA buffer for 1 h. DNA band was visualized on a UV transilluminator and documented with a Gel Logic 212 Pro (Carestream, Woodbridge, CT. USA). The amplicon was purified using a QIAquick Gel Extraction Kit (Qiagen) and the nucleotide sequence was determined in both directions with Taq FS Dye Terminator Cycle Sequencing Fuorescence-Based Sequencing and analyzed on an Applied Biosystems 3730 xl DNA sequencing system The result was an Aspergillus fumisynnematus (figure 5).
Final Diagnosis: Invasive aspergillosis due to A. fumisynnematus infection
Question 2: What treatment is recommended in the care of this patient?
Treatment:
Voriconazole IV, then PO during 8 weeks.
The patient tolerated the treatment adequately, without secondary effects.
Outcome:
Serum galactomannan: <0.5
In the middle of week 7 and 8 we notice a full disappearance of the nodules, and a chest CT revealed recovery of the lung infiltrates.
Discussion:
The genus Aspergillus comprises 344 species and some of these can cause human diseases. Aspergillus fumigatus is a widely distributed mold which has been isolated from natural and residential environment. Infections in the immunocompromised host is often life-threatening [1,2].
Invasive aspergillosis (IA) is most commonly caused by A. fumigatus, amorphospecies identification based on phenotype of colonies and spore-bearing structures. However, an organism previously recognized, as A. fumigatus now constitute a complex that is designated Aspergillus section fumigate subgenus fumigate [3].
Described in 1993 A. fumisynnematus is part of a clade II species from derived from the section Fumigati [4]. Since then A. fumisynnematus has been reported from isolation from the soil and more recently from human samples (bronchoalveolar aspirate). A. fumisynnematus has been described as a separate taxon on the basis of the partial cytochrome b gene sequences of the species [5].
The exposure to azacitidine as part of the treatment for the myelodysplasic syndrome play a key role for the invasive presentation of this Aspergillus species. Previous studies of AZA demonstrated that the absence of antifungal prophylaxis were associated with higher risk of infection [6,7]. Other risk factors for infections are thrombocytopenia and anemia [8] just like what our patient had for prolonged time, thus the invasive aspergillosis probably occurred.
A nice case of a patient with hyper-IgE-syndrome with an invasive aspergillosis related to A. fumisynnematus was described by Mukil Natarajan and colleagues, where a patient manifested elevated IgE level, eosinophilic esophagitis and rhino-orbital-cerebral aspergillosis [9].
Here we present a disseminated aspergillosis related to A. fumisynnematus. We understand that this fungal specie is a rare fungal human pathogen however, the patient was immunocompromised, facilitating his IA infection.
We believe that molecular methods are need for a proper identification of Aspergillus section fumigati to the species level for a better understanding of invasive fungal diseases including drug sensitivity. Unfortunately, we were unable to obtain MICs for this species.
Special thanks to: Monserrat Mayorga-Trejo and Leticia Rojo-Gutiérrez from the Microbiology laboratory at Hospital Médica Sur. Dr. Rigoberto Hernandez-Castro from the research investigation at Hospital General and Dr. Manuel Gea Gonzalez.
Key References:
- Barnes, A.J., Denning, D.W. Aspergilli – Significance as pathogens. Rev Med Microbiol 1993
- Wang L, Yokoyama K, Miyaji M, Nishimura K. Mitochondrial cytochrome b gene analysis of Aspergillus fumigatus and related species. J Clin Microbiol. 2000 Apr;38(4):1352-8
- Vinh DC, Shea YR, Sugui JA, Parrilla-Castellar ER, Freeman AF, Campbell JW, et al. Invasive aspergillosis due to Neosartorya udagawae. Clin Infect Dis. 2009 Jul 1;49(1):102-11.
- Horie Y, Miyaji M, Nishimura K, Taguchi H, Udagawa S. Aspergillus fumisynnematus, a new species from Venezuelan soil. Trans Mycol Soc Jpn. 34:3 (1993)
- Alcazar-Fuoli L, Mellado E, Alastruey-Izquierdo A, Cuenca-Estrella M, Rodriguez-Tudela JL. Aspergillus section Fumigati: antifungal susceptibility patterns and sequence-based identification. Antimicrob Agents Chemother. 2008 Apr;52(4):1244-51
- Lee JH, Lee KH, Lee JH, Kim DY, Kim SH, Lim SN, et al. Decreased incidence of febrile episodes with antibiotic prophylaxis in the treatment of decitabine for myelodysplastic syndrome. Leuk Res. 2011;35:499–503
- Trubiano JA, Dickinson M, Thursky KA, Spelman T, Seymour JF, Slavin MA, et al. Incidence, etiology and timing of infections following azacitidine therapy for myelodysplastic syndromes. Leuk Lymphoma. 2017 Oct;58(10):2379-2386
- Merkel D, Filanovsky K, Gafter-Gvili A, Vidal L, Aviv A, Gatt ME, et al. Predicting infections in high-risk patients with myelodysplastic syndrome/acute myeloid leukemia treated with azacitidine: a retrospective multicenter study. Am J Hematol. 2013;88:130–134.
- Natarajan M, Hsu AP, Weinreich MA, Zhang Y, Niemela JE, Butman JA, et al. Aspergillosis, eosinophilic esophagitis, and allergic rhinitis in STAT3 haploinsufficiency. J Allergy Clin Immunol. 2018; 142:993–997
