Title: Scedosporium CNS Infection in an immunocompetent patient diagnosed by panfungal PCR.
Submitted by: Rita Wilson Dib, MD, Audrey Wanger, PhD,
Luis Ostrosky-Zeichner, MD, Ana V. Salas Vargas, MD,
Institution: University of Texas Health, Houston
Email: rita.wilsondib@uth.tmc.edu
History:
A 36 year old man presented with progressively worsening headache, associated emesis and new onset altered mental status a few days after being discharged following a prolonged and complex hospitalization for cocaine overdose. This recent admission lasted 8 weeks during which he sustained shock with multiorgan failure and hypoxic respiratory failure requiring prolonged mechanical ventilation. His course was also complicated with ventilator associated pneumonia that was appropriately treated.
Other than his active cocaine use, he had a prior history of intravenous drug abuse and alcohol use but did not report events of passing out after drinking. He also had chronic hepatitis C viral infection with prior attempt to treatment that was not completed due to poor compliance.
Physical Examination:
The patient was afebrile with normal vital signs.
The general physical exam was unremarkable, his neurological exam revealed disorientation right sided pronator drift and decreased sensation in his right lower extremity to light touch.
Laboratory Examination:
CBC and CMP were within normal limits. Ammonia, lactic acid and inflammatory markers were not elevated. His urine drug screen was negative.
HIV screen test was not reactive.
Diagnostic Tests Performed:
A CT scan of the head showed 2 cystic masses in the right parietal and left temporal lobe with extensive surrounding vasogenic edema, uncal herniation, and about 6.2 mm left to right midline shift (figure 1). MRI confirmed the presence of multiple intracranial abscesses in the left temporal lobe (5.3 cm) and the right posterior parietal lobe (4.2 cm) without evidence of intraventricular extension (figures 2,3).
The patient underwent left temporal and R parietal craniotomies with evacuation of intracerebral abscesses with notable copious amounts of pus.
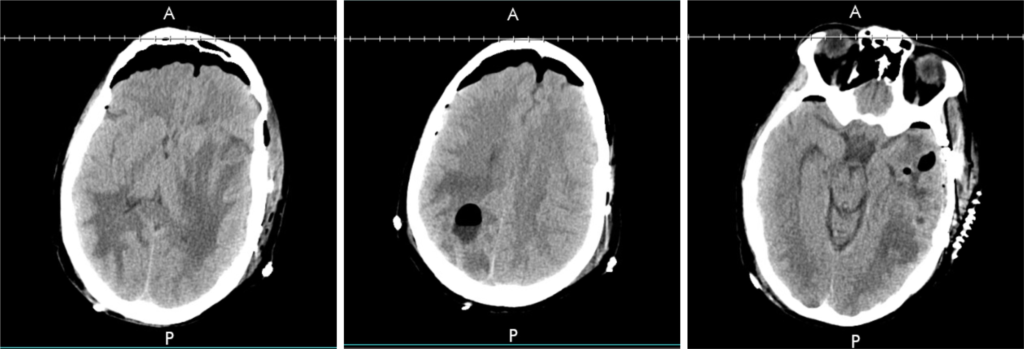
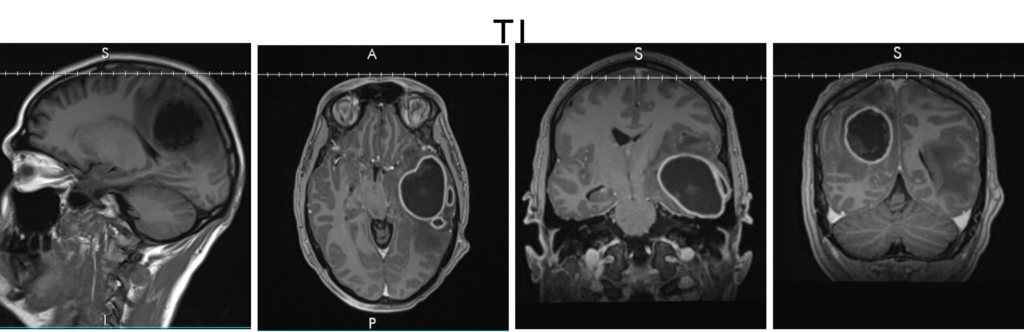
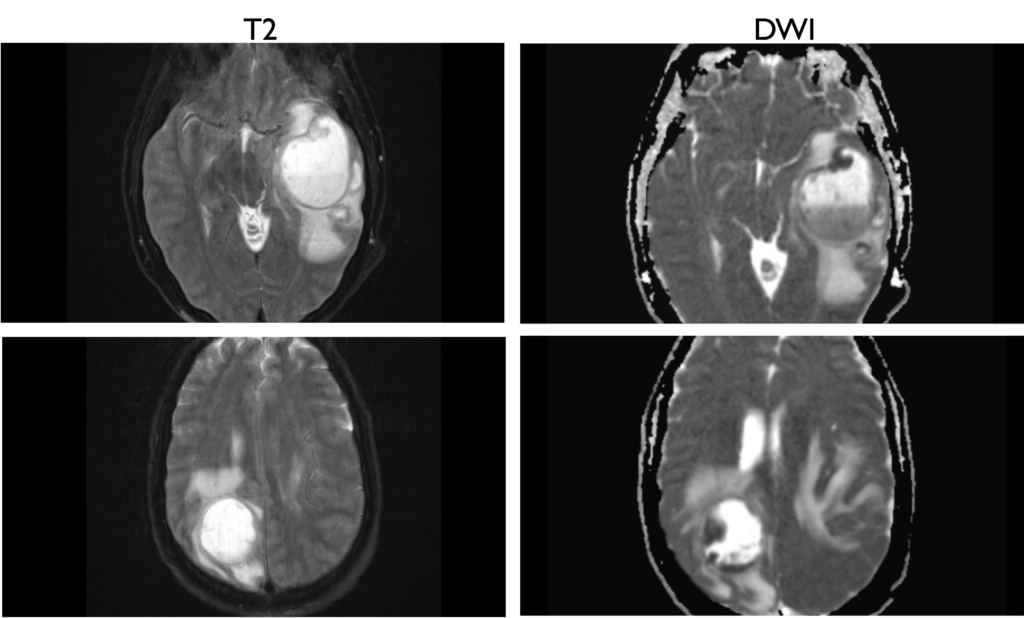
Question 1: What are probable/possible diagnoses?
Microbiology:
Routine bacterial, AFB and fungal stains were unrevealing.
No bacterial growth was noted at 72 hours of incubation. On post-operative day 8, Abscess fluid was then sent for 16S rRNA and Broad range primers fungal PCR which was positive for Scedosporium species.
Fungal cultures eventually were positive for mold which was identified as Scedosporium species by Day 21 post-operatively.
Final Diagnosis:
S. apiospermum Central Nervous System(CNS) abscesses in a non-traditional host
Question 2: What treatment is recommended in the care of this patient?
Treatment:
Aside from surgical evacuation, the patient initially received intravenous voriconazole for 7 days after which, he was switched to oral treatment given his continuous clinical improvement.
Outcome:
A Repeat brain CT 14 days after the initiation of voriconazole revealed Marked interval improvement in the previously noted multiple brain abscesses with residual tiny abscesses in the right parietal lobe and left temporal lobe measuring up to 1.5 cm and 1.8 cm respectively and significant improvement in adjacent vasogenic edema and mass effect with near complete resolution of the rightward midline shift. He continued doing well on clinical follow up 3 months later.
Discussion:
S. apiospermum can cause a wide clinical spectrum of diseases in the immunocompromised population ranging from pulmonary colonization to invasive pulmonary infection, skin and bone infections, and disseminated disease with an apparent neurotropism and high mortality rates [1].
Isolated CNS disease has been reported mainly reported in immunocompetent patients after near drowning in polluted waters [2]. These are thought to occur by contiguous spread from sites near the brain such as the paranasal sinuses or cribriform plate, direct inoculation through a head fracture, Iatrogenic inoculation or hematogenous dissemination from the lungs [3]. One review also noted a small subset of patients (4 out of 99 patients with CNS infections) having a prior history of alcohol abuse [3] while some other cases had no identified predisposition
[4, 5]. Our patient’s initial exposure remained unclear. Assuming his exposure predated his initial admission, the culprit could have been secondary to contaminated cocaine and possible alcoholism.
Species identification is often delayed. The hyphal forms appear as branching with septations and a terminal uninucleate conidia, similar to aspergillus species, making culture and morphological characterization essential for differentiation [1]. In an analysis of 23 cases of CMS scedosporiosis after near drowning, the median time to diagnosis was 28 days from the initial event [6]. This highlights the role PCR-based assays in obtaining an earlier diagnosis and a chance for an early initiation of the adequate therapy in this highly dismal disease [7].
The diagnostic challenges are accompanied by a delay in initiation of appropriate therapy particularly if misidentification as other mold species with different resistance patterns occurs.
Surgical debridement appears to be essential since these infections can be difficult to eradicate with antifungal agents alone [8]. Medical therapy carries its own additional challenges. In vitro and clinical resistance to amphotericin B has been repeatedly documented [9]. Of the triazoles, voriconazole has the best activity in vitro and most reported clinical efficacy observational studies [9]. The success rate reported with voriconazole among patients with CNS involvement is 43% [10].
Posaconazole, Itraconazole and Isavuconazole were noted to have less potent in vitro activity[11, 12]. Echinocandins have variable MECs depending on the S. apiospermum complex species [13] whereas flucytosine and fluconazole have no in vitro activity and terbinafine demonstrated high MICs [10].
Synergy with terbinafine and echinocandins have been reported in vitro although clinical data is limited. Individual case reports suggested some benefit in combination therapy with voriconazole and terbinafine or an echinocandin, amphotericin B with echinocandin [12].
The newer antifungal agents seem to have promising prospects. Olorofim and Fosmanogepix have demonstrated low MICs and MECs respectively against all Scedosporium spp and is currently being evaluated in phase IIb clinical trials [14].
In conclusion, a high index of suspicion and PCR-based diagnostic assays are important to attain an early diagnosis and allow for corresponding earlier adequate management. A medical and surgical approach might carry higher rates of survival. Voriconazole was used successfully in this case, but newer antifungal agents appear to have promising potential.
Key References:
1. Cortez, K.J., et al., Infections caused by Scedosporium spp. Clin Microbiol Rev, 2008. 21(1): p. 157-97.
2. Signore, S.C., et al., Scedosporium apiospermum brain abscesses in a patient after near-drowning – a case report with 10-year follow-up and a review of the literature. Med Mycol Case Rep, 2017. 17: p. 17-19.
3. Kantarcioglu, A.S., J. Guarro, and G.S. de Hoog, Central nervous system infections by members of the Pseudallescheria boydii species complex in healthy and immunocompromised hosts: epidemiology, clinical characteristics and outcome. Mycoses, 2008. 51(4): p. 275-90.
4. Wilson, H.L. and K.J. Kennedy, Scedosporium apiospermum brain abscesses in an immunocompetent man with silicosis. Med Mycol Case Rep, 2013. 2: p. 75-8.
5. Sudke, A.Y., et al., Scedosporium Apiospermum: Rare Cause of Brain Abscess in an Immunocompetent Patient. Neurol India, 2020. 68(4): p. 906-909.
6. Katragkou, A., et al., Scedosporium apiospermum infection after near-drowning. Mycoses, 2007. 50(5): p. 412-21.
7. Lauerer, R.J., et al., Rapid Diagnosis of Central Nervous System Scedosporiosis by Specific Quantitative Polymerase Chain Reaction Applied to Formalin-Fixed, Paraffin-Embedded Tissue. J Fungi (Basel), 2021. 8(1).
8. Jenks, J.D., et al., Clinical characteristics and outcomes of invasive Lomentospora prolificans infections: Analysis of patients in the FungiScope((R)) registry. Mycoses, 2020. 63(5): p. 437-442.
9. Kanafani, Z.A. and J.R. Perfect, Antimicrobial resistance: resistance to antifungal agents: mechanisms and clinical impact. Clin Infect Dis, 2008. 46(1): p. 120-8.
10. Troke, P., et al., Treatment of scedosporiosis with voriconazole: clinical experience with 107 patients. Antimicrob Agents Chemother, 2008. 52(5): p. 1743-50.
11. Meletiadis, J., et al., In vitro activities of new and conventional antifungal agents against clinical Scedosporium isolates. Antimicrob Agents Chemother, 2002. 46(1): p. 62-8.
12. Wiederhold, N.P. and R.E. Lewis, Antifungal activity against Scedosporium species and novel assays to assess antifungal pharmacodynamics against filamentous fungi. Med Mycol, 2009. 47(4): p. 422-32.
13. Lackner, M., et al., Species-specific antifungal susceptibility patterns of Scedosporium and Pseudallescheria species. Antimicrob Agents Chemother, 2012. 56(5): p. 2635-42.
14. Hoenigl, M., et al., The Antifungal Pipeline: Fosmanogepix, Ibrexafungerp, Olorofim, Opelconazole, and Rezafungin. Drugs, 2021. 81(15): p. 1703-1729.
