Background
Nephrotoxicity is one of the more problematic adverse effects of antifungal therapy, specifically with amphotericin B. Although generally reversible, up to 10% of patients with significant kidney dysfunction on amphotericin B will require persistent dialysis after discontinuation of the antifungal [Groll, Piscitelli et al. 1998]. A patient’s risk of developing severe kidney damage during amphotericin B therapy depends on the dose and duration of amphotericin B, underlying health and fluid status of the patient, previous or underlying kidney disease, and the receipt of other potentially nephrotoxic drugs (e.g., aminoglycoside antibiotics, radiocontrast dye, cyclosporine, etc.). For these reasons, the incidence of amphotericin B nephrotoxicity varies widely among different patient populations. For example, a recent retrospective review from a tertiary-care teaching hospital with a sizable oncology and transplant population found that 30% of patients who received amphotericin B deoxycholate developed acute renal failure, resulting in an increased risk of death and prolonged hospital stay [Bates, Su et al. 2001]. However, a similar but smaller study performed at a community hospital without transplant patients found that renal insufficiency occurred in only 15% of patients who received amphotericin B deoxycholate and few if any patients progressed to acute renal failure [Pathak, Pien et al. 1998].
How does nephrotoxicity develop?
Amphotericin B nephrotoxicity can be classified by two mechanisms: 1) the effects of amphotericin B on renal blood flow and glomerular filtration, and 2) the direct toxic effects of amphotericin B on (primarily) the distal tubules.
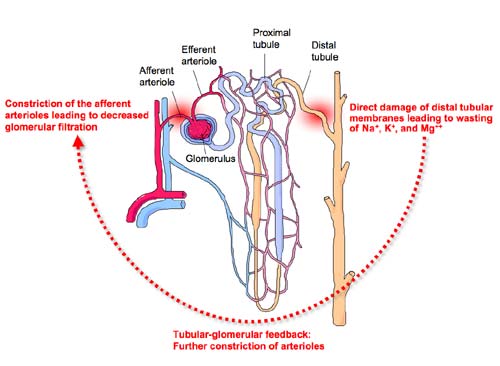
Infusions of amphotericin B result in constriction of the afferent arterioles leading to a drop in renal blood flow and glomerular filtration rate (GFR). Clinically, these effects on the kidney are signaled by an increase in serum creatinine (indicator of GFR) and increasing blood urea nitrogen (azotemia). Glomerular toxicity can develop quickly after a single dose of amphotericin B or evolve slowly after days to weeks of amphotericin B therapy, depending on the hydration status and underlying renal function of the patient. Some clinicians recommend administering intravenous saline before and after amphotericin B infusions, a practice known as sodium loading , to blunt the decreases in the GFR caused by amphotericin B [Branch 1988]. However, the effectiveness of this procedure has not been examined in prospective, controlled clinical trials.
Amphotericin B is also directly toxic to cellular membranes of the distal tubules, resulting in wasting of Na+, K+, and Mg++, impaired urinary acidification and concentration, and eventual renal tubular acidosis [Groll, Piscitelli et al. 1998]. Hypokalemia is typically the first sign of distal tubular toxicity and may require the administration of up to 15 mmol of supplemental potassium per hour to maintain normal serum potassium levels. Hypokalemia and hypomagnesiumemia frequently precede decreases increases in serum creatinine (glomerular damage), especially in patients who are well-hydrated or receiving lipid formulations of amphotericin B. Continued tubular damage, however, eventually causes a decrease in GFR through compensatory tubular-glomerular feedback mechanisms that further constrict the afferent arterioles.
What can be done to reduce nephrotoxicity?
Several approaches have been proposed as strategies to decrease the nephrotoxic effects of amphotericin B therapy. Unfortunately, none of the strategies have been evaluated in well-designed controlled clinical trials, so the relative effectiveness of each approach remains unknown. Vigorous hydration and sodium loading before and after amphotericin B infusions is probably the most accepted strategy for reducing nephrotoxicity, but not all patients can tolerate extra fluids or sodium supplementation. Reducing the daily dosage of amphotericin B or intermittent dosing (e.g., every other day or three times per week) may slow the onset of renal dysfunction, but can potentially compromise the efficacy of antifungal therapy in patients with life-threatening infections. Prolonged or continuous infusions of amphotericin B-deoxycholate (e.g., 1 mg/kg infused over 24 hours) have been reported to be less nephrotoxic [Eriksson, Seifert et al. 2001; Imhof, Walter et al. 2003]. Yet, this dosing approach may not be feasible in patients with limited venous access. Moreover, the efficacy of continuous infusion dosing of amphotericin B has not been well documented.
Probably the best-studied approach for reducing nephrotoxicity with amphotericin B is to administer one of three lipid formulations of amphotericin B (LFAB): ABCD; Amphocil™ or Amphotec™, ABLC; Abelcet™, or L-AMB; Ambisome™. All three LFAB reduce, but do not eliminate nephrotoxicity associated with amphotericin B therapy due (presumably) to their reduced distribution of drug to the kidney [Groll, Piscitelli et al. 1998]. However, the 10-50 fold higher acquisition costs of the LFAB may restricts their initial use to only the highest risk patient populations. For a more detailed discussion of costs associated with these agents, follow this link to the cost pages.
