(described by Link ex Gray in 1821)
Taxonomic Classification
Kingdom: Fungi
Phylum: Ascomycota
Order: Hypocreales
Family: Hypocreaceae
Genus: Fusarium
Description and Natural Habitats
Fusarium is a filamentous fungus widely distributed on plants and in the soil. It is found in normal mycoflora of commodities, such as rice, bean, soybean, and other crops [1806]. While most species are more common at tropical and subtropical areas, some inhabit in soil in cold climates. Some Fusarium species have a teleomorphic state [1295, 2202].
As well as being a common contaminant and a well-known plant pathogen, Fusarium spp. may cause various infections in humans. Fusarium is one of the emerging causes of opportunistic mycoses [63, 66, 531, 916, 1426, 1581, 1826, 1921, 2297, 2304].
Species
The genus Fusarium currently contains over 20 species. The most common of these are Fusarium solani, Fusarium oxysporum, and Fusarium chlamydosporum [531]. Please refer to the table of synonyms for a much more complete list of the currently recognized Fusarium spp.
Synonyms
See the summary of synonyms and teleomorph-anamorph relations for the Fusarium spp.
Pathogenicity and Clinical Significance
As well as being common plant pathogens, Fusarium spp. are causative agents of superficial and systemic infections in humans. Infections due to Fusarium spp. are collectively referred to as fusariosis. The most virulent Fusarium spp. is Fusarium solani [1473]. Trauma is the major predisposing factor for development of cutaneous infections due to Fusarium strains. Disseminated opportunistic infections, on the other hand, develop in immunosuppressed hosts, particularly in neutropenic and transplant patients [142, 306, 844, 2297, 2304]. Fusarium infections following solid organ transplantation tend to remain local and have a better outcome compared to those that develop in patients with hematological malignancies and bone marrow transplantation patients [2015].
Keratitis [581, 1340, 1577, 1963, 2223], endophthalmitis [861, 1374], otitis media [2345], onychomycosis [952, 1961], cutaneous infections [1961] particularly of burn wounds, mycetoma, sinusitis [2041], pulmonary infections [1955], endocarditis, peritonitis, central venous catheter infections, septic arthritis, disseminated infections [63, 66, 531, 916, 1426, 1581, 1826, 1921, 2072], and fungemia [716, 918, 1241, 1248, 2471] due to Fusarium spp. have been reported [531].
Outbreaks of nosocomial fusariosis have also been reported. Existence of Fusarium in hospital water distribution systems may result in disseminated fusariosis in immunosuppressed patients [2141]. Fusarium may also exist in soil of potted plants in hospitals. These plants constitute a hazardous mycotic reservoir for nosocomial fusariosis [2194].
Fusarium spp. produce (A):mycotoxins. Ingestion of grains contaminated with these toxins may give rise to allergic symptoms or be carcinogenic in long-term consumption. Fumonisins are the mycotoxins produced by Fusarium moniliforme and Fusarium proliferatum in maize. They may cause oesophageal cancer [1804]. Another group of mycotoxins, zearalenones, may also be produced by some Fusarium spp. growing in grains [2039]. Studies on reduction or elimination of Fusarium mycotoxins from contaminated agricultural and food commodities are in progress [2334].
Macroscopic Features
Fusarium spp. grow rapidly on Sabouraud dextrose agar at 25°C and produce woolly to cottony, flat, spreading colonies. The only slow-growing species is Fusarium dimerum. From the front, the color of the colony may be white, cream, tan, salmon, cinnamon, yellow, red, violet, pink, or purple. From the reverse, it may be colorless, tan, red, dark purple, or brown.
A sclerotium, which is the organized mass of hyphae that remains dormant during unfavorable conditions, may be observed macroscopically and is usually dark blue in color. On the other hand, sporodochium, the cushion-like mat of hyphae bearing conidiophores over its surface, is usually absent in culture. When present, it may be observed in cream to tan or orange color, except for Fusarium solani, which gives rise to blue-green or blue sporodochia [531, 2202].
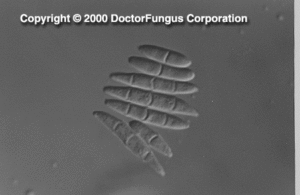
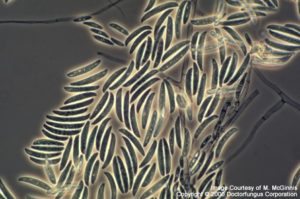
Microscopic Features
Hyaline septate hyphae, conidiophores, phialides, macroconidia, and microconidia are observed microscopically. In addition to these basic elements, chlamydospores are also produced by Fusarium chlamydosporum, Fusarium napiforme, Fusarium oxysporum, Fusarium semitectum, Fusarium solani, and Fusarium sporotrichoides[531, 1295, 2202].
Phialides are cylindrical, with a small collarette, solitary or produced as a component of a complex branching system. Monophialides and polyphialides (in heads or in chains) may be observed. Macroconidia (3-8 x 11-70 µm) are produced from phialides on unbranched or branched conidiophores. They are 2- or more celled, thick-walled, smooth, and cylindrical or sickle- (canoe-)shaped. Macroconidia have a distinct basal foot cell and pointed distal ends. They tend to accumulate in balls or rafts. Microconidia (2-4 x4-8 µm), on the other hand, are formed on long or short simple conidiophores. They are 1-celled (occasionally 2- or 3-celled), smooth, hyaline, ovoid to cylindrical, and arranged in balls (occasionally occurring in chains). Chlamydospores, when present, are sparse, in pairs, clumps or chains. They are thick-walled, hyaline, intercalary or terminal [531, 1295, 2202].
Macroscopic and microscopic features, such as, color of the colony, length and shape of the macroconidia, the number, shape and arrangement of microconidia, and presence or absence of chlamydospores are key features for the differentiation of Fusarium species [531]. Molecular methods, such as 28S rRNA gene sequencing, may be used for rapid identification of Fusarium strains to species level [1024].
Histopathologic Features
See our histopathology page.
Compare to
Acremonium
Cylindrocarpon
Lecythophora
Phialemonium
Fusarium differs from Acremonium, Lecythophora, and Phialemonium by having macroconidia. It differs from Cylindrocarpon by having macroconidia with foot cells and pointed distal ends [2202].
Laboratory Precautions
No special precautions other than general laboratory precautions are required.
Susceptibility
Fusarium is one of the most drug-resistant fungi. Among the Fusarium spp., Fusarium solani in general tends to be most resistant of all. Fusarium strains yield quite high MICs for flucytosine, ketoconazole, miconazole, fluconazole, itraconazole, and posaconazole [119, 1434, 1854, 1899, 2282, 2283]. The novel triazole, Syn-2869, has no activity against Fusarium [1131]. Fusarium spp. are intrinsically resistant to the novel glucan synthesis inhibitors, caspofungin, anidulafungin, and micafungin [120, 558, 683, 1780, 2226]. Despite the lack of its activity alone, the combination of caspofungin with amphotericin B appears synergistic against some Fusarium isolates [121].
The only antifungal drugs that yield relatively low MICs for Fusarium are amphotericin B [65, 1899, 1973], voriconazole [446, 687, 1152, 1433, 1494, 1864, 2432], and natamycin [1899, 1973]. Compared to itraconazole, voriconazole yields notably lower MICs [119, 1130]. Terbinafine may show good in vitro activity against some isolates [2136].
For MICs of various antifungal drugs for Fusarium, see our N/A(L):susceptibility database.
Fusarium infections are difficult to treat and the invasive forms are often fatal. Amphotericin B alone or in combination with flucytosine or rifampin is the most commonly used antifungal drug for treatment of systemic fusariosis [2015]. Lipid formulations of amphotericin B, such as liposomal amphotericin B [455] and amphotericin B lipid complex [2370, 2452] are also used. However, most cases remain resistant and fail to respond to amphotericin B treatment. Granulocyte and GM-CSF transfusions concommitant to amphotericin B therapy may be life-saving in some immunosuppressed patients with disseminated fusariosis [2137]. Despite its limited in vitro activity, posaconazole appears effective in murine fusariosis [1380]. Human data are awaited for verification of this finding.
Topical natamycin is used for treatment of keratitis due to Fusarium [1866]. In addition to antifungal therapy, keratoplasty is required for several patients [2111]. Patients with mycetoma due to Fusarium may respond to itraconazole [1895]. Onychomycosis due to Fusarium, on the other hand, may be treated with itraconazole and ciclopirox nail lacquer.